Siberian Ginseng est planta. Populus uti radix de plant facere Medicine.siberian ginseng est saepe vocavit per "Adaptogen." Hoc est a non-medical terminus solebat describere substantiae, quod potest uelut confirma corpus et crescere generalis resistentiam ad cotidie stress.in praeter quod usus est ad ADTAPTOGEN, Siberian Ginseng adhibetur ad condiciones et in corde et sanguine vasa, talis est usus ad conditionibus, humilis sanguinem, et talis est usus ad condiciones, humilis sanguinem, tanem in arteriis (ATHEROSCLEROSIS), Horrenibus in Arteriis (ATHEROSCLEROSIS), Horrening De Arteriis (ATHEROSCLEROSIS) et High Sanguis (ATHEROSCLEROSIS), et RHEUMATICUS CORDEUS.
Product Name: Siberian Ginseng Extract
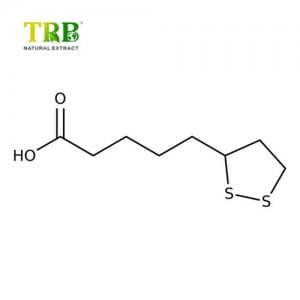
Latine nomen: Eleutherocus senticosus (Rupr.et Maxim.) Nocet
CAS non: 7374-79-0
Plant Part Used: Rhizome
Asay: Eleutheroside B + E 0,8%, 1.5%, 2.0% per HPLC
Color: brunneis pulveris cum propria odor et gustum
GMO Status: GMO Free
Packing: in 25kgs fibra tympanis
PRAECLUSIO: Custodite continens unopined in frigus, sicco locus, ut auferat a fortis lux
FLUTEM Vita: XXIV menses a date ex productio
Function:
-Anti-lassitudine; Anti-inflammatione; Augendae corpus resistentia contra morbum.
-Expel ventus et uliginem, confirma tendinum.
-Nourish Cor et Tranquilize mens: nam defectus cordis manifestatur ut insomnia, somnia et palpitatio; Et tractare mare morbi vel adversa reactionem ad altum habitus vel sub humilis temperatus aut altum aqua.
-Benefit vitalis industria, quia lien-defectus cum pauper appetitus, solutam advenerit et lassitudine. Nuper, etiam propter leukocytopenia, coronarius cor morbo, longos BRONCHITIS, ThromboGiITIS obliterans, aut ut subsidiary anti-carcinogenic pharmaca aut X-ray Lorem ad tumorem.
Application:
-As rudis materiae medicinae, quod maxime in pharmaceutical agri;
-Functional cibo ut Capsulae vel diripio;
-Health products ut Capsulae vel diripio.
Technical notitia sheet
| Item | Specificatio | Ratio | Res |
| Idem | PRAEFECTIO | N / est | Obsequium |
| Extract solvents | Aqua / Ethanol | N / est | Obsequium |
| Particula magnitudine | C% transitum LXXX reticulum | USP / Ph.eur | Obsequium |
| Mole density | 0.45 ~ 0.65 g / ml | USP / Ph.eur | Obsequium |
| Damnum siccatio | ≤5.0% | USP / Ph.eur | Obsequium |
| Sulpatsa | ≤5.0% | USP / Ph.eur | Obsequium |
| Plumbum (PB) | ≤1.0mg / kg | USP / Ph.eur | Obsequium |
| Arsenicum (ut) | ≤1.0mg / kg | USP / Ph.eur | Obsequium |
| Cadmium (Paperback) | ≤1.0mg / kg | USP / Ph.eur | Obsequium |
| Solvents residuum | USP / Ph.eur | USP / Ph.eur | Obsequium |
| Pesticides residui | Negans | USP / Ph.eur | Obsequium |
| Microbiological imperium | |||
| Otal bacterial comitem | ≤1000cfu / g | USP / Ph.eur | Obsequium |
| Fermentum & fingunt | ≤100cfu / g | USP / Ph.eur | Obsequium |
| Salmonella | Negans | USP / Ph.eur | Obsequium |
| E.coli | Negans | USP / Ph.eur | Obsequium |
| More Information of TRB | ||
| Certification ordinatione | ||
| USFDA, CEP, Kosher Halal Gmp ISO testimoniales | ||
| RELLILIUM | ||
| Fere XX annis, export XL terris et regiones, magis quam MM batches produci a trb non aliqua qualitas problems, unique purification processus, in inmunditia et castitate imperium in occursum USP et CP | ||
| Comprehendo qualitas system | ||
|
| ▲ quality securitate ratio | √ |
| ▲ Document imperium | √ | |
| ▲ Ratio Validation | √ | |
| ▲ disciplina ratio | √ | |
| ▲ Internum Audit Protocol | √ | |
| ▲ suppler audit system | √ | |
| ▲ apparatu facilities ratio | √ | |
| ▲ Materia Imperium System | √ | |
| ▲ Lorem imperium ratio | √ | |
| ▲ Packaging Labeling System | √ | |
| ▲ Laboratory Imperium System | √ | |
| ▲ verificationem validation system | √ | |
| ▲ Regulatory Negotiis Ratio | √ | |
| Imperium totum fontibus et processibus | ||
| Strictly regi omnes rudis materia, accessiones et packaging materiales.prereferred rudis materiae et accessiones et packaging materiae elit nobiscum DMF numerus.Pluribus rudis materia amet ut copiam certare. | ||
| Fortis cooperantem institutions ad firmamentum | ||
| Institutum Botany / Institution of Microbiology / Academy of Science and Technology / University | ||