Product Name:Yohimbine HCL 98.0%
Other Name:Yohimbine HCL ; Yohimbe HCl ; 11-hydroxy Yohimbine, Alpha Yohimbine HCl, Coryanthe Yohimbe, Corynanthe Johimbe, Corynanthe johimbi, Corynanthe yohimbi, Johimbi, Pausinystalia yohimbe, Pausinystalia johimbe, Yohimbehe, Yohimbehe Cortex, Yohimbine, , Yohimbinum Muriaticum.
Botanical Source:yohimbe bark extract
Part: Bark(Dried, 100% Natural)
Extraction Method: Water/ Grain Alcohol
Form:white crystalline powder
Specification: 98%
Test Method : HPLC
CAS Number:146-48-5/65-19-0
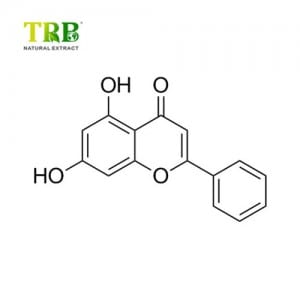
Molecular Formula:C21H26N2O3
Molecular Weight: 354.45
GMO Status:GMO Free
Packing: in 25kgs fiber drums
Storage:Keep container unopened in cool, dry place,Keep away from strong light
Shelf Life:24 months from date of production
Yohimbine HCl 98.0% Product Description
Product Overview
Yohimbine Hydrochloride (CAS No.: 65-19-0) is a highly purified alkaloid derived from the bark of the African plant Pausinystalia yohimbe. With a molecular formula of C21H26N2O3·HCl and a molecular weight of 390.91 g/mol, this compound is provided as a white to off-white crystalline powder, ensuring consistent quality for research and pharmaceutical applications. The product meets stringent purity standards of ≥98.0% as verified by HPLC analysis .
Key Specifications
- Purity: ≥98.0% (HPLC)
- Appearance: White crystalline powder
- Melting Point: 288–290°C
- Specific Optical Rotation: +100° to +105° (H2O)
- Solubility: Soluble in DMSO, methanol, ethanol; slightly soluble in water
Quality Assurance
- Analytical Methods: Compliant with USP 35 and European Pharmacopoeia standards, including:Certification: Supported by a Certificate of Analysis (CoA) confirming batch-specific purity and stability .
- HPLC for purity quantification
- Infrared Absorption Spectroscopy (IR) and UV Spectrophotometry for structural verification
- Impurity Control: Limits specified for impurities A, F, G (general use) and B, C, D, E (pharmaceutical grade)
Applications
- Research Use: Studied for its role as a selective α2-adrenergic receptor antagonist .
- Pharmaceutical Intermediate: Utilized in controlled formulations under regulatory compliance (e.g., FDA-approved prescription drugs for erectile dysfunction) .
- Non-Medical Note: Not intended for human consumption, dietary supplements, or veterinary use due to potential health risks from unregulated dosing .
Safety & Handling
- Hazard Classification: Acute toxicity (oral, dermal, inhalation; GHS Category 1) .
- Precautionary Measures:Storage: Store in a cool, dry place (2–8°C), protected from light and moisture .
- Use protective gloves and avoid inhalation/contact with skin/eyes .
- In case of exposure: Rinse thoroughly with water and seek medical attention .
Packaging & Logistics
- Available Sizes: 1 kg (aluminum foil bag), 25 kg (fiber drum) .
- Shelf Life: 2 years under recommended storage conditions .
- Transport Compliance: Classified as UN 1544 (6.1 Toxic Substance, Packing Group II) .
Regulatory Compliance
- Research-Only Statement: Exclusively for laboratory use; not evaluated for diagnostic/therapeutic purposes .
- Global Standards: Meets USP, EP, and OSHA HCS requirements for chemical safety .
Why Choose This Product?
- Global Sourcing: Manufactured under ISO-certified facilities with traceable supply chains .
- Technical Support: Comprehensive CoA and chemical characterization data (1H-NMR, HPLC) provided .
- Customization: Available in varying purities (2%–98%) and formulations upon request .
Keywords : Yohimbine HCl 98%, CAS 65-19-0, Alpha-2 Adrenergic Blocker, Pharmaceutical Grade, Research Chemical, HPLC Verified, USP Compliance.